Abstract
Klebsiella pneumoniae recombinants convert glycerol to the special chemical 1,3-propanediol (1,3-PDO), which is used for the production of polytrimethylene terephthalate (PTT). In this study, a NAD(+)-dependent gamma-glutamyl-gamma-aminobutyraldehyde dehydrogenase (PC) was cloned from K. pneumoniae DSM 2026, which oxidizes 3-hydroxypropionaldehyde to a chemical platform of 3-hydroxy propionic acid (3-HP ). and overexpressed in K. pneumonia DSM 2026 for co-production of 3-HP and 1,3-PDO from glycerol.
In addition, the dhaT gene, encoding NADH-dependent 1,3-propanediol oxidoreductase (1,3-PDOR), was deleted from the chromosome for the balanced production of 3-HP and 1,3-PDO. Recombinant K. pneumoniae ∆dhaT, expressing pure, produced 3.6 g 3-HP and 3.0 g 1,3-PDO per litre with a mean yield of 81% in flask glycerol charcoal culture stirring under microaerobic conditions.
When fed-batch culture was carried out under microaerobic conditions at pH 7.0 in a 5 L bioreactor, the recombinant K. pneumoniae ∆dhaT(plus) strain produced 16.0 g 3-HP and 16.8 g of 1,3-PDO per litre with a cumulative yield of 51% on glycerol carbon in 24 h. The production of 1,3-PDO in the dhaT deletion mutant was attributed to hypothetical NAD(P)H-dependent oxidoreductase expression. This study demonstrates the feasibility of obtaining two commercially valuable chemicals, 3-HP and 1,3-PDO, on a significant scale.
Product specifications
- Size: 10ug, 50ug, 100ug or 500ug
- Format: Liquid
- Formulation: Supplied as a filtered 0.45 um solution in 100 mM NaH2PO4 (pH 7.4), 0.3 M NaCl, 4 M urea.
- Purification: His-tag affinity purification by immobilized metal ion affinity chromatography (IMAC)
- Concentration: Lot Specific (see label on the vial)
- Purity: >95%
- Endotoxin Level: Not determined
- Purity determined by: SDS-PAGE under reducing conditions and visualized by Coomassie blue staining
- Preservative: None
- Recommended Applications: ELISA, EIA, Protein-Protein Interaction Studies, Western Blot, Dot Blot, Immunoprecipitation, Protein Array
Isolation and identification of bacteria
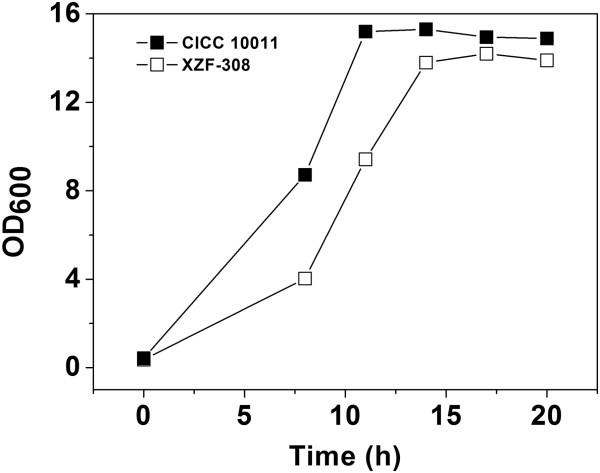
K. pneumoniae R46 was isolated on MacConkey agar (MCA) plate containing florfenicol (8 mg/L) from an anal swab sample from a rabbit farm in Wenzhou, South China. Species identification was performed with the Vitek-60 self-assessment system for microorganisms (BioMerieux Corporate, Lyon, France). Further verification was performed using homology comparisons of the 16S ribosomal RNA gene and the NCBI nucleotide database whole-genome sequence via the BLASTN software. Multilocus sequence typing (MLST) was performed by analyzing the housekeeping genes gapA, info, mdh, pgi, phoE, rpoB and tone through the online service MLST-1.8.
Antimicrobial susceptibility tests
The agar dilution method was used to determine the minimum inhibitory concentrations (MIC) of antibiotics against bacteria according to the guidelines of the Clinical and Laboratory Standards Institute (CLSI document M100-S28, 2017) and the European Committee on Antimicrobial Susceptibility Testing. (EUCAST 2017). All tests were performed in triplicate. Briefly, each isolate was diluted in the logarithmic growth period with 0.9% saline to achieve a standard suspension of 0.5 McFarland, equivalent to an inoculum.
Suspensions of approximately 5 μL were seeded on Mueller-Hinton (MH) agar plates (with a series of antibiotic concentrations between 0.0625 and 1024 mg/L). The inoculated plates were then incubated for 18 hours at 37°C in a constant temperature incubator. The MIC was defined as the lowest antibiotic concentration that showed no colony growth. The commercially available E. coli strain ATCC 25922 served to ensure the quality of the plates, and E. coli DH5α carrying an empty cloning vector was used for comparison.
Conjugation experiment
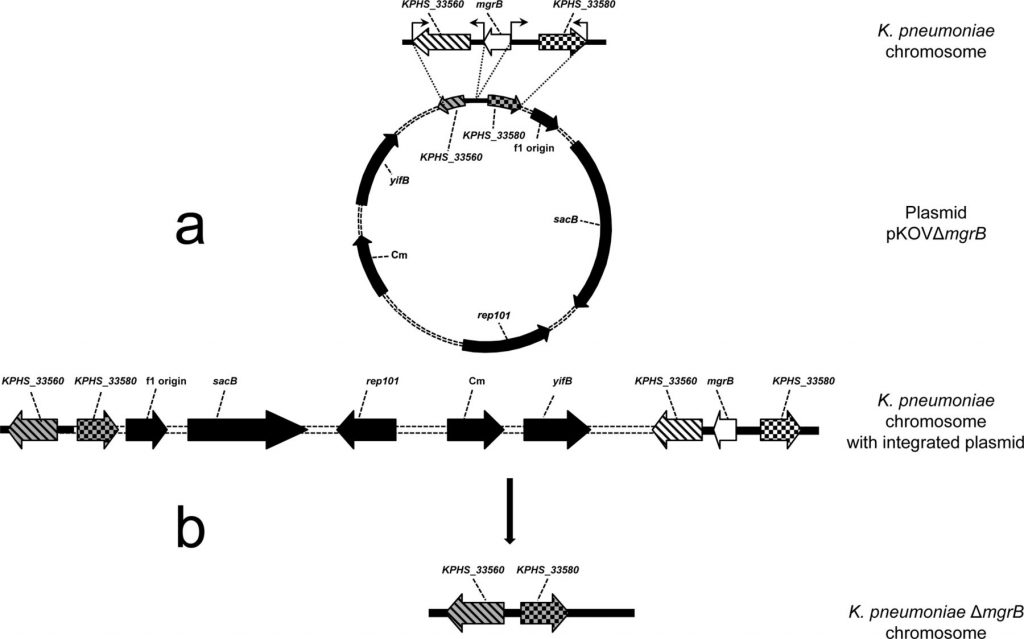
A conjugation experiment was carried out by biparental mating on sterile nitrocellulose filters and rifampicin-resistant E. coli C600 (EC600) was used as recipient. Overnight cultures of donor strain K. pneumoniae R46 and recipient strain EC600 were mixed in 3 mL LB broth, harvested, and resuspended in 80 μL LB. The mixture was placed on a 1 cm 2 filter membrane, plated on an LB plate, and then incubated for mating at 37°C for 12-18 hours.
Transconjugants were selected on two types of Mueller-Hinton agar plates: one containing 1,024 mg/L rifampin and 32 mg/L florfenicol, and the other containing 1,024 mg/L rifampin and 32 mg/L chloramphenicol. Plasmid DNA from the transconjugant was extracted and analyzed by agarose gel electrophoresis and compared to the plasmid profile of K. pneumonia R46. PCR analysis of the genes encoded on the plasmid was performed to further verify the plasmid in the transconjugants.
Fountain
- Genetic symbols: BLAKPC-2
- Species: Klebsiella pneumonia
- Access number: W5TZT6
- Expressed region: Leu25-Gln293
- Protein name and synonyms: BlaKPC-2, KPC-2
Comparative Genomic Analysis
Sequences containing resistance genes were obtained from the NCBI Nucleotide Database by the BLASTN program using the K. pneumoniae R46 resistance gene sequences as queries. The resulting sequences were filtered and only sequences containing resistance genes were kept. CD-HIT was used to pool the retained sequences using the K. pneumoniae R46 genome sequence as a reference with 90% identity and 85% coverage. The sequence that shared the greatest similarity with the other sequences in each group was chosen as a candidate for orthologous analysis. Orthologous groups of genes to candidate sequences were identified using BLASTp. Sequence retrieval, statistical analysis, and other bioinformatics tools used in this study were applied with Perl and Bioperl scripts.
Preparation
- Expression System: E.Coli
- Conjugation/Tag: Histidine tag 6x N-terminal
- Molecular weight (kDa): 31.1
Nucleotide accession numbers
The sequences are now available in the NCBI Nucleotide Database. The accession numbers for the chromosome and plasmids are CP035777, CP035774, CP035775 and CP035776, respectively.
Stability and Storage
Shipping: Ice packs
Storage/Stability
Upon arrival, protein can be stored for 2 weeks at 4°C. For long-term storage, it is recommended to store at -20°C or -80°C in suitable aliquots. Avoid repeated cycles of freezing and thawing.